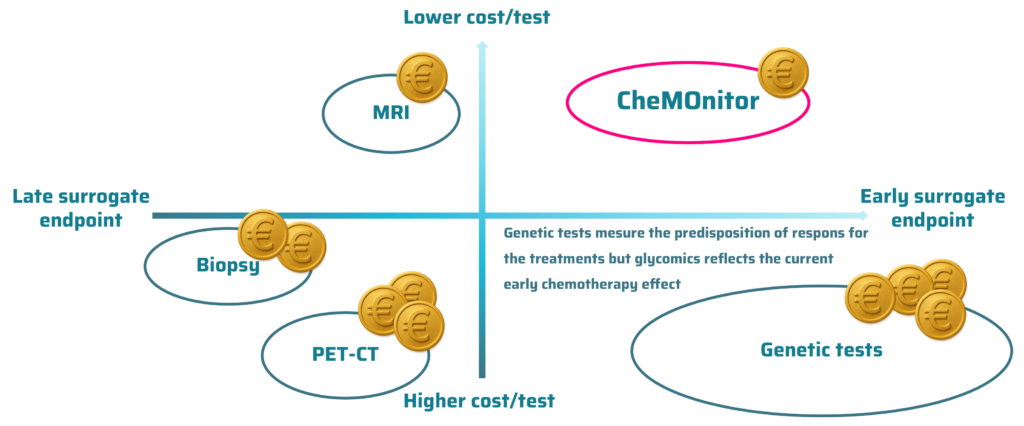
About CheMOnitor
The problem
Oncologist cannot predict the effectiveness of chemotherapy
1. Protocol-based chemotherapy selection
Oncologists follow standard treatment guidelines — yet only ̴ 30% of chemotherapy regimens prove effective in lung cancer patients
2. Delayed treatment feedback
Current imaging techniques require up to 3 months to assess responseà late treatment-correction à lowers recovery chance
3. High cost of ineffective treatment
Unsuccessful chemotherapy results in unnecessary drug expenses, additional diagnostics, and avoidable interventions à estimated therapy cost: ̴ €12,000/patient
4. Strain on healthcare systems
Prolonged hospitalization à 20-25 days/patient/year + repeated treatments place a heavy burden on oncology services and staff
5. Reduced patient quality of life and economic productivity
Side effects from ineffective treatments worsen quality of life and lead to lost workdays à ̴ 100 days/patient/year, and increased societal costs

The solution
Our technology predicts the effectiveness of chemotherapy after the first dose
1. Early screening for ineffective chemotherapy
Our test enables early detection of ineffective treatments — after the first dose of chemotherapy
2. Lower cost from avoiding ineffective treatments1,2,3
Avoids ineffective therapies and treatment related cost à saving ̴ €6,000/patient
3. Improved 5-year survival rate
Timely treatment adjustment leads to significantly better long-term patient outcomes
4. Improved patient quality of life
Enhances quality of life, reducing physical and emotional burden
5. Time savings and higher productivity
Reduces hospitalization days and increases economic productivity
Our vision
We strive to transform the landscape of lung cancer care by becoming the first to offer a molecular diagnostic tool that can predict chemotherapy effectiveness—empowering clinicians to make earlier, more informed treatment decisions. With over 1.4 million patients receiving chemotherapy for lung cancer each year and no current solution addressing this diagnostic gap, we see a vital opportunity to bring precision oncology to the forefront. Our vision is to become a key player in this evolving field, starting in the EU and USA, where personalized medicine is gaining momentum.
By offering both direct diagnostic services and scalable reagent kits, we aim to make our solution accessible, cost-effective, and impactful. Looking ahead, we are committed to capturing at least 1% of the EU market by 2035, driving better outcomes for patients and greater efficiency for healthcare systems.
Competitive advantage
- First adaptation of an emerging technology in the oncology domain
- Patent pending (#P2400268)
- Solid scientific background – published papers
- Tested on 100 samples with 92% accuracy
- Unique combination of cutting-edge analytics with AI driven data interpretation
- Diverse team (scientists, engineers, business professionals, AI experts, and oncologists)
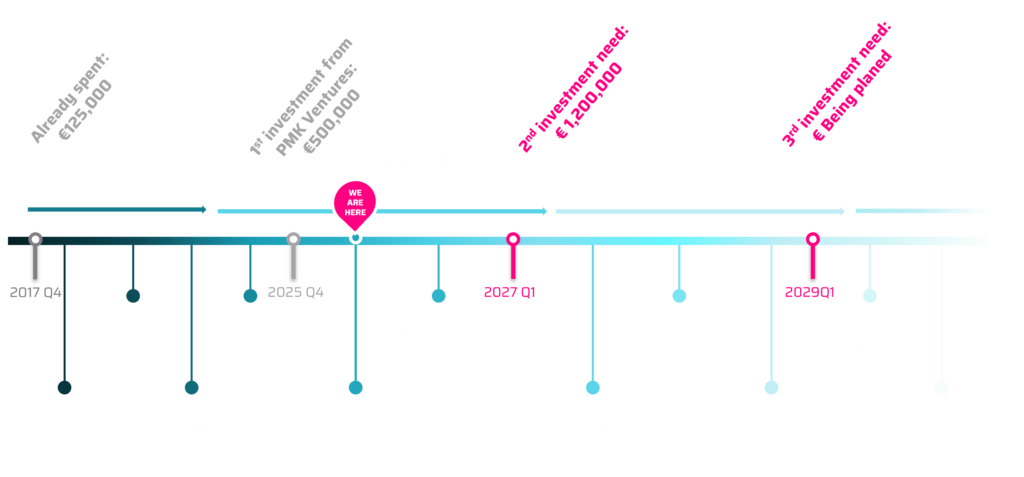
Key milestones
Our implementation plan follows a structured timeline with key milestones.
- The project started in 2018, and by 2024, we finalized the proof of concept.
- In 2025, we established CheMOnitor Ltd. has secured its first €500,000 investment round to accelerate technology development, scheduled for completion by Q2 2027.
- The second funding round (€1.2M) in 2027 will support regulatory approval, including CE marking in 2028.
- The final investment, currently being planned, will enable the market launch in 2029.
We mitigate these through early stakeholder engagement, continuous R&D improvements, and a diversified investment strategy.
Market potential
Lung cancer is the second most common cancer and the leading cause of cancer deaths worldwide. Each year, approximately 300,000 new cases are registered in the European Union and 61% of the patients are treated with chemotherapy.
By improving the prediction of chemotherapy effectiveness, we can improve treatment outcomes and reduce healthcare costs. The substantial number of patients requiring effective therapeutic solutions indicates a growing market with considerable opportunities for impact and expansion.
“Business potential – Not a screening, not a prevention… it’s a need! “
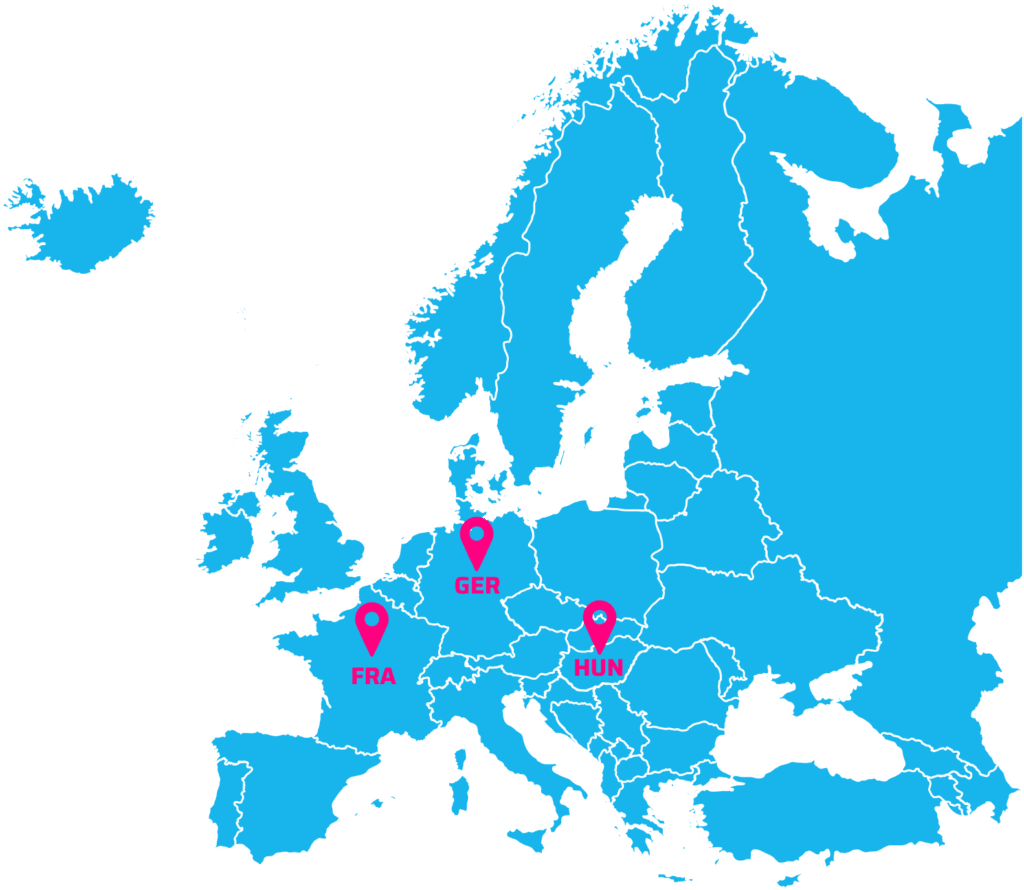
Hungary: 10 500 lung cancer patients > € 6,5M / year
Germany: 67 000 lung cancer patients > € 50M / year
France: 54 000 lung cancer patients > € 48M / year
Why invest in CheMOnitor?
CheMOnitor is contributing to the field of lung cancer therapy with its innovative solutions. Investing in our company means backing a breakthrough in personalized cancer treatment, with the potential to save lives and significantly reduce healthcare costs, offering substantial returns and market impact.
Behind the technology
IP status
Our proprietary method, currently under patent pending status (#P2400268), combines ultra-high resolution glycan analysis with advanced machine learning to predict chemotherapy effectiveness in lung cancer patients.
The Scientific Basis of the Technology
- Vathy-Fogarassy, A., et al., Improved analytical workflow towards machine learning supported N-glycomics-based biomarker discovery, Talanta, 2025
- Torok, R., et al., Predicting the effectiveness of chemotherapy treatment in lung cancer utilizing artificial intelligence-supported serum N-glycome analysis, Computers in Biology and Medicine, 2025
- Torok, R., et al., The Effect of Sample Glucose Content on PNGase F-Mediated N-Glycan Release Analyzed by Capillary Electrophoresis, Molecules, 2022
- Torok, R., et al., Evaluation of Possible Processing Time Effects on the Global N-Glycosylation Profile of Human Blood Samples, Current Molecular Medicine, 2021
- Mészáros, B., et al., Comparative analysis of the human serum N-glycome in lung cancer, COPD and their comorbidity using capillary electrophoresis. Journal of Chromatography B, 2020
- Mészáros, B., et al., Machine Learning Based Analysis of Human Serum N-glycome Alterations to Follow up Lung Tumor Surgery, Cancers, 2020
- Reider, B., et al., Evaporative fluorophore labeling of carbohydrates via reductive amination, Talanta, 2018